8 Year Exporter Blood Glucose Monitoring Device - UBREATH ® Spirometer System (PF680) – e-Linkcare
8 Year Exporter Blood Glucose Monitoring Device - UBREATH ® Spirometer System (PF680) – e-Linkcare Detail:

Measurable Spirometry through Inhale & Exhale
FVC, SVC, MVV are available with 23 parameters to be calculated.
Accuracy and repeatability comply with ATS/ERS task force standardization (ISO26782:2009)
Complies with the ATS/ERS requirement for flow sensitivity down to 0.025L/s which is a vital characteristic for the diagnosis and monitoring of COPD patients.
Real-time Graphic Curve Experience
Synchronized graphs enhance users to achieve satisfied results with professional’s guidance.
Displayed three waveform parameters and remark the best performance for reference.
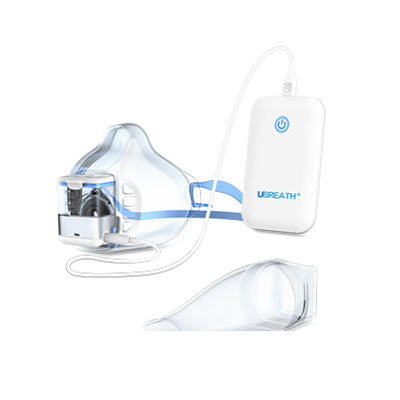
Portable Design
Hand-held device and easy to operation.
Automated BTPS calibration and free of environmental conditions influence.
Lightweight combines the benefits of portability.
Operate with Safety
Assured hygiene with disposable pneumotach gives NO authority to cross-contamination.
Patented design offers prevention.
Automated quality control and correction algorithm to minimize interference from operation.
All-in-One Service Station
Built-in printer and barcode scanner are combined in one device.
LIS/HIS connection via Wi-Fi and HL7.
Technical Specifications
| Feature | Specification |
| Model | PF680 |
| Parameter | FVC: FVC, FEV1, FEV1%, PEF, FEF25, FEF50, FEF75VC: VC, VT, IRV, ERV, IC MVV: MVV, VT, RR |
| Flow Detection Principle | Pneumotachograph |
| Volume Range | Volume: (0.5-8) LFlow: (0-14) L/s |
| Performance Standard | ATS/ERS 2005 & ISO 26783:2009 |
| Volume Accuracy | ±3% or ±0.050L (take the larger value) |
| Power Supply | 3.7 V lithium battery (rechargeable) |
| Printer | Built-in thermal printer |
| Operating Temperature | 10℃ - 40℃ |
| Operating Relative Humidity | ≤ 80% |
| Size | Spirometer: 133x82x68 mmSensor Handle: 82x59x33 mm |
| Weight | 575g (including the Flow Transducer) |
Product detail pictures:

Related Product Guide:
It adheres for the tenet "Honest, industrious, enterprising, innovative" to develop new products and solutions continuously. It regards shoppers, success as its individual success. Let us produce prosperous future hand in hand for 8 Year Exporter Blood Glucose Monitoring Device - UBREATH ® Spirometer System (PF680) – e-Linkcare , The product will supply to all over the world, such as: Singapore, Pakistan, Kazakhstan, Welcome to visit our company, factory and our showroom where displays various hair products that will meet your expectation. Meanwhile, it is convenient to visit our website, and our sales staff will try their best to provide you the best service. Please contact us if you need more information. Our aim is to help customers realize their goals. We are making great efforts to achieve this win-win situation.
This company has the idea of "better quality, lower processing costs, prices are more reasonable", so they have competitive product quality and price, that's the main reason we chose to cooperate.