FeNO in Respiratory Diagnostics: A Modern Approach to Airway Inflammation Assessment
Introduction
Asthma affects approximately 262 million people worldwide and remains one of the most common chronic respiratory diseases, causing significant morbidity and placing substantial burden on healthcare systems. For decades, the diagnosis and management of asthma have relied heavily on symptom assessment and spirometry—tools that measure lung function but do not directly evaluate the underlying airway inflammation that drives the disease. Enter Fractional exhaled Nitric Oxide (FeNO) , a simple, non-invasive breath test that is transforming respiratory medicine by providing direct, real-time insight into Type 2 airway inflammation.

What Is FeNO and Why Does It Matter?
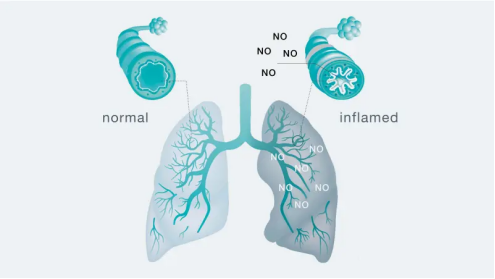
Nitric oxide (NO) is a gaseous signaling molecule naturally produced by the epithelial cells lining the airways. Under normal conditions, low levels of NO are present in exhaled breath. However, when the airways become inflamed—particularly in response to allergic triggers—these cells dramatically increase NO production. This NO then diffuses into the airway lumen and is exhaled, where it can be measured as FeNO.
FeNO serves as a direct, non-invasive biomarker for eosinophilic (Type 2) airway inflammation, the predominant inflammatory pathway in allergic asthma. This inflammation is driven by cytokines such as interleukin-4, -13, and -5, and is characterized by elevated immunoglobulin E (IgE), eosinophils in blood and sputum, and responsiveness to inhaled corticosteroids (ICS).
The clinical value of FeNO lies in its ability to provide objective, quantifiable data about airway inflammation that would otherwise remain invisible. Unlike spirometry, which assesses airflow obstruction, FeNO directly measures the inflammatory process itself, offering complementary information that enhances diagnostic accuracy and guides treatment decisions.
A Paradigm Shift: The 2025 GINA Guidelines
The release of the 2025 GINA guidelines marks a pivotal moment in respiratory diagnostics. For the first time, FeNO has been formally expanded beyond its traditional role in disease management to now actively support the diagnosis of Type 2 asthma.
The guidelines now recommend integrating FeNO testing into the initial diagnostic workup when asthma is suspected and access to the test is available. According to GINA 2025, if a patient presents with typical asthma symptoms but lung function testing is not available or yields normal results, an elevated FeNO value can support the diagnosis of Type 2 asthma.
The guidelines provide clear cut-off values indicative of Type 2 asthma: ≥50 ppb for adults and ≥35 ppb for children who are ICS-naïve. For patients already on treatment, the thresholds are lower: ≥25 ppb for those using medium-dose ICS, and ≥20 ppb for those using high-dose ICS.
This elevation of FeNO to a diagnostic tool acknowledges the central role of phenotyping in modern asthma care and enables a more precise, biologically grounded approach to initial diagnosis.
Clinical Applications of FeNO Testing
Diagnosis of Asthma
FeNO testing excels in differentiating Type 2-high asthma from other conditions that may present with similar symptoms, such as chronic cough, vocal cord dysfunction, or non-T2 asthma phenotypes. A consistently elevated FeNO level in a patient with suggestive symptoms provides compelling positive evidence for a Type 2-high endotype, even before a trial of treatment.
In the United Kingdom, FeNO testing has been recommended as part of the NICE asthma diagnosis guideline, and an estimated additional 102,704 patients across England have benefited from FeNO testing through national programs.
Predicting Treatment Response
A high FeNO level strongly predicts favorable response to inhaled corticosteroids (ICS) and Type 2-targeted biologic therapies. This allows clinicians to stratify therapy more rationally from the outset, facilitating a personalized, "right-first-time" treatment approach that can improve early control and outcomes.
Monitoring Disease Control and Guiding Treatment Adjustments
Serial FeNO measurements provide objective assessment of treatment adherence and the suppression of underlying Type 2 inflammation. An elevated FeNO level despite prescribed ICS therapy often indicates poor medication adherence, enabling healthcare providers to address this without confrontation. FeNO trends can also inform decisions on stepping up or stepping down ICS dosage.
Predicting Exacerbation Risk
FeNO is also included in the GINA guidelines for assessing a patient's risk of exacerbation, even when symptom control appears good. This predictive capability enables earlier intervention and potentially prevents acute attacks.
Beyond Asthma: Other Respiratory Applications
While asthma remains the primary application, FeNO testing has demonstrated value in chronic cough (differentiating allergic from other causes), chronic obstructive pulmonary disease (identifying asthma-COPD overlap), allergic rhinitis, and eosinophilic bronchitis.
The Future of FeNO in Precision Respiratory Medicine
The integration of FeNO into mainstream respiratory diagnostics is accelerating. The 2025 GINA guidelines have embedded FeNO testing across diagnosis, assessment, and management, recognizing it as a key biomarker for identifying Type 2 inflammation and personalizing treatment. As precision medicine advances, FeNO is increasingly being used to guide biologic therapy selection for severe asthma, particularly for agents targeting the IL-4/IL-13 pathway.
The UBreath BA200 is ideally positioned to support this evolution. With its high-precision electrochemical sensor, rapid turnaround time, and comprehensive measurement capabilities, the device enables healthcare providers to implement FeNO-guided care pathways in both primary and secondary care settings. Its portable design and intuitive interface make it accessible for point-of-care use in clinics, hospitals, and even remote healthcare facilities.
Conclusion
FeNO testing has fundamentally changed the landscape of respiratory medicine by providing a simple, non-invasive window into airway inflammation. From supporting the diagnosis of Type 2 asthma to guiding treatment decisions and monitoring disease control, this breath test offers invaluable objective data that enhances patient care.
The UBreath BA200 represents the state-of-the-art in FeNO diagnostic technology. With its high-precision electrochemical sensing, comprehensive multi-parameter measurement capabilities, rapid results, and user-friendly design, the UBreath BA200 empowers healthcare professionals to deliver more accurate, personalized, and effective respiratory care. Backed by international clinical guidelines and designed for long-term reliability, the UBreath BA200 is an essential tool for any clinical practice committed to excellence in respiratory diagnostics.
As asthma and other chronic respiratory diseases continue to pose significant challenges to public health worldwide, embracing innovations like the UBreath BA200 represents a step forward in early diagnosis, optimized treatment strategies, and ultimately, better quality of life for patients.