Tuning Insulin Receptor Signaling with AI-Designed Agonists: A New Class of Diabetes Therapy That Mitigates Cancer Risk
Since the discovery of insulin by Banting a century ago, it has been a cornerstone in the treatment of diabetes. However, therapies based on insulin and its analogs have several limitations, such as complex production processes, stringent storage requirements, potential promotion of cancer cell proliferation, and ineffectiveness in some patients with hereditary insulin resistance.
Insulin functions by activating the Insulin Receptor (IR) on the cell surface. This receptor acts like a complex "lock," and insulin serves as the "key" to unlock it. Upon activation of the insulin receptor, two primary signaling pathways are initiated: one regulates metabolism (the AKT pathway), responsible for lowering blood glucose, while the other regulates cell growth (the MAPK pathway), which may promote cancer cell proliferation. Dysregulation of insulin receptor signaling can therefore lead to diseases such as diabetes and cancer.

Leveraging AI-based de novo protein design, it is possible to create ultra-stable, easily producible proteins that function as insulin receptor agonists, mimicking the role of insulin. This approach holds promise for overcoming the current limitations of insulin and its analogs.
On October 13, 2025, a research team including Professor David Baker and Dr. Wang Xinru from the University of Washington's Institute for Protein Design, and Researcher Bai Xiaochen from the University of Texas Southwestern Medical Center, published a study titled "Tuning insulin receptor signaling using de novo-designed agonists" in Molecular Cell, a subsidiary journal of Cell.
This study utilized AI-based de novo protein design to generate insulin receptor agonists (AI insulin). These not only demonstrated superior blood glucose-lowering effects compared to insulin but also enabled precise regulation of in vivo signaling pathways, avoiding the promotion of cancer cell growth by insulin. This lays the foundation for developing more effective and safer diabetes treatments.
In this latest study, the research team—comprising scientists from the University of Washington, the University of Texas Southwestern Medical Center, and Columbia University—adopted a novel strategy. Moving beyond the constraints of natural insulin's structure, they employed AI-based de novo protein design to generate entirely new insulin receptor agonists.
The team first designed two binding proteins: one specifically binding to the L1 domain (site-1, the primary insulin-binding site) of the insulin receptor, and the other binding to the FnIII-1 domain (site-2, a secondary insulin-binding site). These two binding proteins were then connected via linkers of varying lengths to form novel insulin receptor agonist molecules.
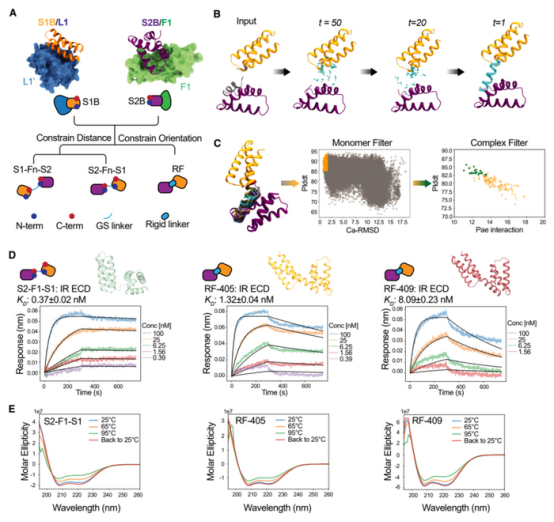
These designed insulin receptor agonists exhibited remarkable properties:
Enhanced Thermal Stability: Unlike natural insulin, the newly designed proteins remained stable even at 95°C, significantly simplifying storage and transportation requirements.
Precise Signaling Control: By adjusting the rigidity of the linkers, the research team achieved precise control over the signaling pathways. Designs with rigid linkers (e.g., RF-405) activated both the metabolic (AKT) and cell growth (MAPK) pathways, whereas designs with flexible linkers (e.g., S2-F1-S1) primarily activated the metabolic pathway, substantially reducing stimulation of cell growth.
In mouse experiments, these designed insulin receptor agonist molecules demonstrated significant advantages. For instance, RF-409 was more effective than insulin at lowering blood glucose, achieving the same effect at half the dose and with a longer duration of action. In an obese diabetic mouse model, a single injection of RF-409 maintained low blood glucose levels for up to 6 hours, whereas blood glucose began to rise within 2 hours after insulin treatment.
Notably, these designed agonists were able to activate genetically mutated insulin receptors in some patients with insulin resistance, offering new therapeutic hope for rare hereditary forms of diabetes.
Most encouragingly, while these designed insulin receptor agonist molecules effectively activated the insulin receptor in normal cells, they failed to do so in breast cancer cells. Further investigation revealed that this was because cancer cells co-express the insulin receptor and the IGF-1 receptor, readily forming hybrid receptors. The designed agonists specifically bind only to the insulin receptor.
This specific binding capability suggests that these designed agonists could significantly reduce the potential cancer risk associated with traditional insulin therapy. This is particularly beneficial for patients requiring long-term insulin treatment who are concerned about cancer risks.
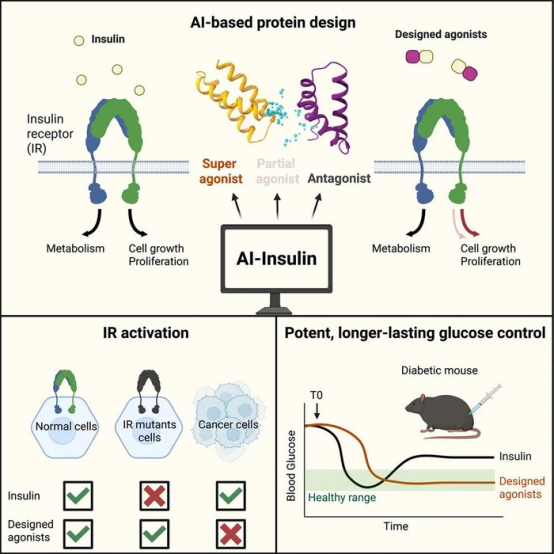
In summary, while natural insulin activates the insulin receptor in normal cells but fails to activate mutated receptors and may activate the receptor in cancer cells, the AI-designed de novo insulin receptor agonists can activate the insulin receptor in normal cells, activate mutated receptors, and avoid activation in cancer cells. Additionally, the designed agonists offer more sustained and stable blood glucose-lowering effects.
This study not only demonstrates how to design safer and more effective insulin receptor agonists but also elucidates the fundamental relationship between receptor activation and signal transduction, laying the groundwork for the development of safer and more effective next-generation diabetes therapeutics.
Even as diabetes treatment options become increasingly innovative and effective, the most critical aspect remains diligent blood glucose monitoring and staying attuned to one's own body in real-time. This foundational practice is the cornerstone of effective diabetes management, enabling timely adjustments and ensuring optimal outcomes alongside advanced therapies.

